Scoping call
30 minYou share the program context and the question. We define what you’ll get and when.
Quinn, the AI scientist, synthesizes biological, clinical, and competitive evidence around your drug asset and delivers a structured answer your team can defend internally.
Target validation · Indication prioritization · Biomarker strategy · Trial design · Any therapeutic area, any modality
Discuss your programTrusted by teams at








You have a program decision coming up, and the evidence to make it well exists across published literature, expression databases, trial registries, and competitive intelligence. But pulling it together into something your team can defend takes months of manual work.
The decision won’t wait. So it gets made on conviction and committee dynamics. Not because the data isn’t there, but because nobody had time to synthesize it.
Each deliverable maps to a decision your team is making. Not a landscape scan. Not a slide deck. A structured answer.
A ranked shortlist your governance committee can act on. Scored across biological plausibility, competitive landscape, and clinical feasibility, with documented rationale for every inclusion and exclusion.
An independent assessment of whether your target and modality hold up. Druggability, tractability, freedom-to-operate, mechanism, and competitive position.
The patient subgroups most likely to respond, identified from genomic, proteomic, and clinical evidence. Implementable selection logic, not a research summary.
Adaptive design, endpoint strategy, sample size, and comparator rationale. Grounded in your program’s evidence and ready for protocol discussion.
These are the most common starting points. The methodology adapts to any stage, indication, or modality.
Discovery
Target ID · MoA elucidation · Indication expansion
Preclinical
Biologics design · Lead optimization · CMC
Clinical
Trial design · Biomarker strategy · Endpoints
Regulatory
IND gap analysis · Safety summaries
Commercial
TPP & valuation · Competitive intel
Ranked targets with tractability, freedom-to-operate, rationale, disease model, and validation protocols.
Two selection rules ready to implement. Filed into the protocol.
A ranked shortlist your governance committee can act on.
You share the program context and the question. We define what you’ll get and when.
We run the analysis independently, from public evidence or your proprietary data. No integration, no IT involvement on your side.
A structured deliverable with traceable findings, reproducible code, validation protocols, and a scientific briefing with your team.
Most teams start with one question on one asset — then come back.
A program team needed to rank candidate indications for a novel biologic before their next governance discussion. Quinn delivered a scored shortlist in 5 days across biological plausibility, competitive landscape, and clinical feasibility, with documented rationale for every inclusion and exclusion. Three indications advanced.
A clinical program team needed independent verification of a target’s biological rationale before committing to a Phase II. Quinn integrated genetic evidence, expression data, and competitive intelligence in 4 days. The target held up and a previously unreported biomarker opportunity was identified.
A program team was about to lock a protocol without knowing if their biomarker subgroup was real. Quinn resolved the biomarker into four distinct states and delivered two selection rules in 3 days. They went straight into the filing strategy.
Every engagement produces a structured evidence package tied to the specific decision your team is making. Here’s the shape of what you receive.
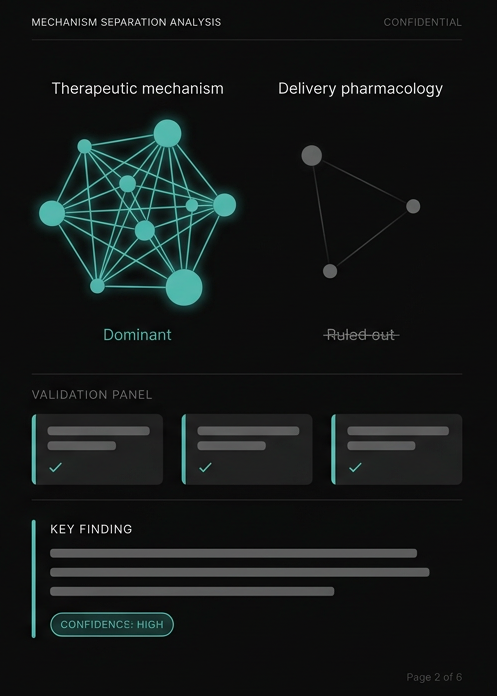
Is the early post-dose response in our Phase I study driven by delivery pharmacology or therapeutic mechanism? And can we validate the answer on existing specimens?
Construct modification as the response driver. Single-cell RNA-seq for the separation question. Module-level transcriptional rankings.
A 3-feature panel discriminated two response patterns across repeated sensitivity testing. Early responses were delivery-dominant. The signal resolved within 24 hours.
3 days. Equivalent internal path: 5–7 months.
The team knew which lever to pull before committing to either path. The panel validated immediately on existing specimens.
TRACEABLE TO SOURCE · REPRODUCIBLE CODE · METHODOLOGY DOCUMENTED · AUDIT-READY
Scientists who’ve done this work inside pharma R&D. Stanford spinout, backed by Y Combinator. Co-founded by Daniel Gomari, PhD (Snyder Lab, Stanford) and Michael Snyder, PhD (Chair of Genetics, Stanford).
Your IP stays yours · Never trains on your data · SOC 2 Type II · ISO 27001 · HIPAA compliant
If there’s a fit, we’ll scope the work on a 30-minute call.
Or reach us directly at team@quinnbio.com